Secondary Metabolites in Plants

Secondary metabolites are chemicals produced by plants for which no role has yet been found in growth, photosynthesis, reproduction, or other "primary" functions. These chemicals are extremely diverse; many thousands have been identified in several major classes. Each plant family, genus, and species produces a characteristic mix of these chemicals, and they can sometimes be used as taxonomic characters in classifying plants. Humans use some of these compounds as medicines, flavorings, or recreational drugs.
Secondary metabolites can be classified on the basis of chemical structure (for example, having rings, containing a sugar), composition (containing nitrogen or not), their solubility in various solvents, or the pathway by which they are synthesized (e.g., phenylpropanoid, which produces tannins). A simple classification includes three main groups: the terpenes (made from mevalonic acid, composed almost entirely of carbon and hydrogen), phenolics (made from simple sugars, containing benzene rings, hydrogen, and oxygen), and nitrogen-containing compounds (extremely diverse, may also contain sulfur).
The apparent lack of primary function in the plant, combined with the observation that many secondary metabolites have specific negative impacts on other organisms such as herbivores and pathogens , leads to the hypothesis that they have evolved because of their protective value. Many secondary metabolites are toxic or repellant to herbivores and microbes and help defend plants producing them. Production increases when a plant is attacked by herbivores or pathogens. Some compounds are released into the air when plants are attacked by insects; these compounds attract parasites and predators that kill the herbivores. Recent research is identifying more and more primary roles for these chemicals in plants as signals, antioxidants , and other functions, so "secondary" may not be an accurate description in the future.
Consuming some secondary metabolites can have severe consequences. Alkaloids can block ion channels, inhibit enzymes , or interfere with neurotransmission, producing hallucinations , loss of coordination, convulsions, vomiting, and death. Some phenolics interfere with digestion, slow growth, block enzyme activity and cell division, or just taste awful.
Most herbivores and plant pathogens possess mechanisms that ameliorate the impacts of plant metabolites, leading to evolutionary associations between particular groups of pests and plants. Some herbivores (for example, the monarch butterfly) can store (sequester) plant toxins and gain protection against their enemies. Secondary metabolites may also inhibit the growth of competitor plants (allelopathy). Pigments (such as terpenoid carotenes, phenolics, and flavonoids) color flowers and, together with terpene and phenolic odors, attract pollinators.
Secondary chemicals are important in plant use by humans. Most pharmaceuticals are based on plant chemical structures, and secondary metabolites are widely used for recreation and stimulation (the alkaloids nicotine and cocaine; the terpene cannabinol). The study of such plant use is called ethnopharmacology. Psychoactive plant chemicals are central to some religions, and flavors of secondary compounds shape our food preferences. The characteristic flavors and aroma of cabbage and relatives are caused by
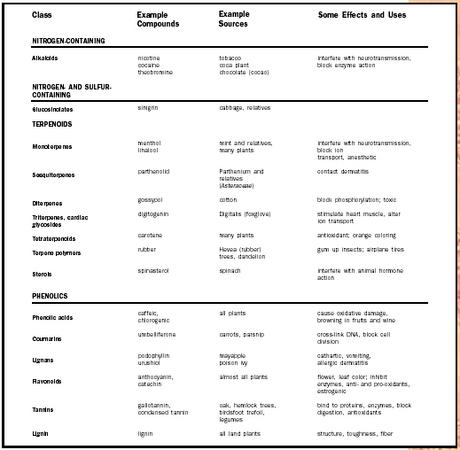
| Class | Example Compounds | Example Sources | Some Effects and Uses |
| NITROGEN-CONTAINING | |||
| Alkaloids | nicotine cocaine theobromine | tobacco coca plant chocolate (cocao) | interfere with neurotransmission, block enzyme action |
| NITROGEN-AND SULFUR-CONTAINING | |||
| Glucosinolates | sinigrin | cabbage, relatives | |
| TERPENOIDS | |||
| Monoterpenes | menthol linalool | mint and relatives, many plants | interfere with neurotransmission, block ion transport, anesthetic |
| Sesquiterpenes | parthenolid | Parthenium and relatives ( Asteraceae ) | contact dermatitis |
| Diterpenes | gossypol | cotton | block phosphorylation; toxic |
| Triterpenes, cardiac glycosides | digitogenin | Digitalis (foxglove) | stimulate heart muscle, alter ion transport |
| Tetraterpenoids | carotene | many plants | antioxidant; orange coloring |
| Terpene polymers | rubber | Hevea (rubber) trees, dandelion | gum up insects; airplane tires |
| Sterols | spinasterol | spinach | interfere with animal hormone action |
| PHENOLICS | |||
| Phenolic acids | caffeic, chlorogenic | all plants | cause oxidative damage, browning in fruits and wine |
| Coumarins | umbelliferone | carrots, parsnip | cross-link DNA, block cell division |
| Lignans | podophyllin urushiol | mayapple poison ivy | cathartic, vomiting, allergic dermatitis |
| Flavonoids | anthocyanin, catechin | almost all plants | flower, leaf color; inhibit enzymes, anti- and pro-oxidants, estrogenic |
| Tannins | gallotannin, condensed tannin | oak, hemlock trees, birdsfoot trefoil, legumes | bind to proteins, enzymes, block digestion, antioxidants |
| Lignin | lignin | all land plants | structure, toughness, fiber |
nitrogen-and sulfur-containing chemicals, glucosinolates, which protect these plants from many enemies. The astringency of wine and chocolate derives from tannins. The use of spices and other seasonings developed from their combined uses as preservatives (since they are antibiotic) and flavorings.
SEE ALSO Flowers ; Herbivory and Plant Defenses ; Metabolism, Cellular ; Poisons
Jack Schultz
Bibliography
Agosta, William. Bombardier Beetles and Fever Trees: A Close-up Look at Chemical Warfare and Signals in Animals and Plants. Reading, MA: Addison-Wesley, 1996.
Bidlack, Wayne R. Phytochemicals as Bioactive Agents. Lancaster, PA: Technomic Publishers, 2000.
Karban, Richard, and Ian T. Baldwin. Induced Responses to Herbivory. Chicago: University of Chicago Press, 1997.
Rosenthal, Gerald A., and May R. Berenbaum. Herbivores, Their Interactions with Secondary Plant Metabolites. San Diego, CA: Academic Press, 1991.