Ion Channels

Ions are charged particles such as Na + , H + , K + , Ca 2+ , and Cl - . Ions have a significant effect on many cell processes and also influence the amount of water in the cell. Cells use inorganic ions for transmitting signals across the cell membrane or along the surface of the cell. Other cellular functions as diverse as secretion of hormones to fertilization of egg cells require ion transport across the cell membrane. However, ions have great difficulty passing through the membrane by simple diffusion because cell membranes are composed of hydrophobic phospholipids that oppose the passage of hydrophilic ions. Furthermore, the negatively charged phosphate head groups of the phospholipids tend to repel negatively charged anions and trap positively charged cations. Therefore, an ion as small as a hydrogen ion (H + ) requires a specific portal protein to facilitate its transport through the membrane. Such a protein molecule is called an ion channel.
Molecular Structure of Potassium and Sodium Channels
An ion channel is usually equipped with four basic parts: a central conduction pathway (opening) for ions to pass through, an ion recognition site to allow passage of specific ions (selectivity filter), one or more gates that may open or close, and a sensor that senses the triggering signal and transmits it to the gate.
The Shaker-type voltage-gated potassium channel of nerve and muscle provides a good example of the four parts of the ion channel. The name Shaker arises from the gene coding for this channel in the fruitfly ( Drosophila melanogaster ), whose mutation causes the fly to shake. Humans have many potassium channels belonging to the Shaker family. This channel is composed of four identical subunits arranged like a four-leaf clover, with the center serving as the ion conduction pathway. Each subunit has six segments that cross the membrane and are termed S1 through S6. The region between S5 and S6 segments from each subunit contributes to form the ion conduction pathway; hence, it is called the "pore" or "Pregion."
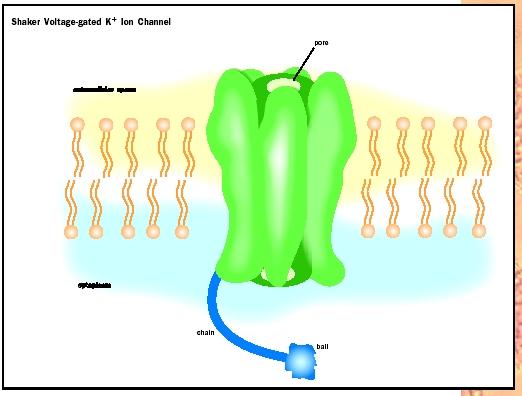
In the P-region, a few critical amino acids from each subunit gather to form the selectivity filter that specifically recognizes only potassium ions. The S4 segment contains positively charged amino acids on every third position and serves as a voltage sensor. When the potential on the internal surface of the membrane becomes more positive, the potential drives the S4 segment toward the outside. This movement triggers a channel gate to open. The voltage-gated sodium channel has a similar architecture, except that the four subunits are strung together in a long peptide chain like a train of parading elephants linking up trunk-to-tail. This channel is highly selective for sodium ions.
Biophysics
As the charged ions flow across the membrane, they generate an electric current. The amount of current flow is determined by three factors. First, when the gate of an ion channel opens, ions flow down the concentration gradient from high to low across the membrane, which is typical of the passive transport mechanism. Second, the flow of ions is controlled by the voltage difference across the membrane. For instance, if the cell interior is already highly positive, less K + will flow in. Third, a channel may be highly selective for a specific ion (such as the voltage-gated sodium channel) or rather nonselective (such as the mechano-sensitive channel). Thus, the total ion flow is influenced by the concentration gradient of the ions, the voltage difference across the membrane, and the permeability of the ions.
The patch clamp technique developed in 1980 has enabled scientists to record current flow through a single ion channel. This technique uses extremely fine glass electrodes attached to membranes to measure electrical activity in a very small part of the membrane. One of the most exciting results from the development of the patch clamp technique is direct observation of the opening and closing of a single channel, like observing the twinkle of a little star in the night sky. The opening of a channel represents a conformational change of the channel molecule from a closed state to an open state. If the rate for such a conformational change is dependent on voltage, then the channel is said to be voltage-gated. A channel may stay in the open state for less than a millisecond to tens of seconds. The current flow through a single channel may range from less than a picoampere to hundreds of picoamperes (a picoampere is 10 -12 ampere).
Drugs and Toxins Acting on Ion Channels
Nature produces a wide variety of highly potent toxins that target specific ion channels. The toxins are usually packaged in venom and delivered by stings or fangs. A large number of toxins have been isolated from scorpions, sea anemones, cone snails, and snakes. They have been used for studying various ion channels. One of the most famous toxins is tetrodotoxin, which selectively blocks the sodium channel. It is contained in the poisonous puffer fish, which ironically is the most expensive delicacy served in Japanese restaurants. Only chefs who have passed rigorous licensing examinations are allowed to prepare the fish. Tetrodotoxin is also commonly portrayed in fictions and movies; it almost killed the fictitious Agent 007 James Bond in From Russia with Love. Drugs have been developed to target ion channels and to prevent the channels from conducting ions. They are widely used as local anesthetics, antiarrhythmic drugs to prevent irregular heartbeats, antihypertensive drugs to lower blood pressure, and anti-epileptic drugs to prevent seizures.
Genetic Defects of Ion Channels
Several genetic diseases exhibiting defects in the physiological functions of ion channels have now been shown to be caused by mutations in the genes coding for specific ion channels. For example, a cardiac potassium channel named HERG (human ether-a-go-go-related gene) acts to protect the heart against inappropriate rhythmicity. People lacking a functional HERG gene exhibit an abnormality on their electrocardiogram called "long Q-T syndrome," which predisposes them to sudden cardiac arrest when they are under stress. Cystic fibrosis results from mutations of a particular chloride channel called the cystic fibrosis transmembrane conductance regulator.
SEE ALSO Membrane Proteins ; Membrane Transport ; Neuron ; Synaptic Transmission
Chau H. Wu
Bibliography
Hille, Bertil. Ionic Channels of Excitable Membranes, 2nd ed. Sunderland, MA: Sinauer Associates Inc., 1992.
Neher, Erwin, and Bert Sakmann. "The Patch Clamp Technique." Scientific American 266 (1992): 44–51.
A first taste to what might be great!
Thank you,
Edward