Reproduction in Plants

Plant reproduction is the process by which plants generate new individuals, or offspring. Reproduction is either sexual or asexual. Sexual reproduction is the formation of offspring by the fusion of gametes . Asexual reproduction is the formation of offspring without the fusion of gametes. Sexual reproduction results in offspring genetically different from the parents. Asexual offspring are genetically identical except for mutation. In higher plants, offspring are packaged in a protective seed, which can be long lived and can disperse the offspring some distance from the parents. In flowering plants (angiosperms), the seed itself is contained inside a fruit, which may protect the developing seeds and aid in their dispersal.
Sexual Reproduction in Angiosperms: Ovule Formation
All plants have a life cycle that consists of two distinct forms that differ in size and the number of chromosomes per cell. In flowering plants, the
The flower produces two kinds of gametophytes, male and female. The female gametophyte arises from a cell within the ovule , a small structure within the ovary of the flower. The ovary is a larger structure within the flower that contains and protects usually many ovules. Flowering plants are unique in that their ovules are entirely enclosed in the ovary. The ovary itself is part of a larger structure called the carpel, which consists of the stigma, style, and ovary. Each ovule is attached to ovary tissue by a stalk called the funicle. The point of attachment of the funicle to the ovary is called the placenta.
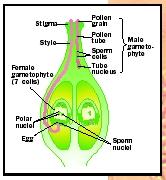
As the flower develops from a bud, a cell within an ovule called the archespore enlarges to form an embryo-sac mother cell (EMC). The EMC divides by meiosis to produce four megaspores. In this process the number of chromosomes is reduced from two sets in the EMC to one set in the megaspores, making the megaspores haploid. Three of the four megaspores degenerate and disappear, while the fourth divides mitotically three times to produce eight haploid cells. These cells together constitute the female gametophyte, called the embryo sac.
The eight embryo sac cells differentiate into two synergids, three antipodal cells, two fused endosperm nuclei, and an egg cell. The mature embryo sac is situated at the outer opening (micropyle) of the ovule, ready to receive the sperm cells delivered by the male gametophyte.
Pollen
The male gametophyte is the mature pollen grain. Pollen is produced in the anthers, which are attached at the distal end of filaments. The filament and anther together constitute the stamen, the male sex organ. Flowers usually produce many stamens just inside of the petals. As the flower matures, cells in the anther divide mitotically to produce pollen mother cells (PMC). The PMCs divide by meiosis to produce haploid microspores in groups of four called tetrads. The microspores are housed within a single layer of cells called the tapetum, which provides nutrition to the developing pollen grains.
Each microspore develops a hard, opaque outer layer called the exine, which is constructed from a lipoprotein called sporopollenin. The exine has characteristic pores, ridges, or projections that can often be used to identify a species, even in fossil pollen. The microspore divides mitotically once or twice to produce two or three haploid nuclei inside the mature pollen grain. Two of the nuclei function as sperm nuclei that can eventually fuse with the egg and endosperm nuclei of the embryo sac, producing an embryo and endosperm, respectively.
For sexual fusion to take place, however, the pollen grain must be transported to the stigma, which is a receptive platform on the top of the style, an elongated extension on top of the carpel(s). Here the moist surface or chemicals cause the pollen grain to germinate. Germination is the growth of a tube from the surface of a pollen grain. The tube is a sheath of pectin , inside of which is a solution of water, solutes , and the two or three nuclei, which lack any cell walls. Proper growth of the pollen tube requires an aqueous solution of appropriate solute concentration, as well as nutrients such as boron, which may aid in its synthesis of pectin.
At the apex of the tube are active ribosomes and endoplasmic reticulum (types of cell organelles ) involved in protein synthesis. Pectinase and a glucanase (both enzymes that break down carbohydrates ) probably maintain flexibility of the growing tube and aid in penetration. The pollen tube apex also releases ribonucleic acid (RNA) and ribosomes into the tissues of the style. The tube grows to eventually reach the ovary, where it may travel along intercellular spaces until it reaches a placenta. Through chemical recognition, the pollen tube changes its direction of growth and penetrates through the placenta to the ovule. Here the tube reaches the embryo sac lying close to the micropyle, and sexual fertilization takes place.
Double Fertilization
Fertilization in flowering plants is unique among all known organisms, in that not one but two cells are fertilized, in a process called double fertilization. One sperm nucleus in the pollen tube fuses with the egg cell in the embryo sac, and the other sperm nucleus fuses with the diploid endosperm nucleus. The fertilized egg cell is a zygote that develops into the diploid embryo of the sporophyte. The fertilized endosperm nucleus develops into the triploid endosperm, a nutritive tissue that sustains the embryo and seedling. The only other known plant group exhibiting double fertilization is the Gnetales in the genus Ephedra, a nonflowering seed plant. However, in this case the second fertilization product degenerates and does not develop into endosperm.
Double fertilization begins when the pollen tube grows into one of the two synergid cells in the embryo sac, possibly as a result of chemical attraction to calcium. After penetrating the synergid, the apex of the pollen tube breaks open, releasing the two sperm nuclei and other contents into the synergid. As the synergid degenerates, it envelops the egg and endosperm cells, holding the two sperm nuclei close and the other expelled contents of the pollen tube. The egg cell then opens and engulfs the sperm cell, whose membrane breaks apart and allows the nucleus to move near the egg nucleus. The nuclear envelopes then disintegrate, and the two nuclei combine to form the single diploid nucleus of the zygote. The other sperm cell fuses with the two endosperm nuclei, forming a single triploid cell, the primary endosperm cell, which divides mitotically into the endosperm tissue.
Double fertilization and the production of endosperm may have contributed to the great ecological success of flowering plants by accelerating the growth of seedlings and improving survival at this vulnerable stage. Faster seedling development may have given flowering plants the upper hand in competition with gymnosperm seedlings in some habitats, leading to the abundance of flowering plants in most temperate and tropical regions. Gymnosperms nevertheless are still dominant at higher elevations and latitudes, and at low elevations in the Pacific Northwest coniferous forests, such as the coastal redwoods. The reasons for these patterns are still controversial.
The Seed
The seed is the mature, fertilized ovule. After fertilization, the haploid cells of the embryo sac disintegrate. The maternally derived diploid cells of the ovule develop into the hard, water-resistant outer covering of the seed, called the testa, or seed coat. The diploid zygote develops into the embryo, and the triploid endosperm cells multiply and provide nutrition. The testa usually shows a scar called the hilum where the ovule was originally attached to the funicle. In some seeds a ridge along the testa called the raphe shows where the funicle originally was pressed against the ovule. The micropyle of the ovule usually survives as a small pore in the seed coat that allows passage of water during germination of the seed.
In some species, the funicle develops into a larger structure on the seed called an aril, which is often brightly colored, juicy, and contains sugars that are consumed by animals that may also disperse the seed (as in nutmeg, arrowroot, oxalis, and castor bean). This is distinct from the fruit, which forms from the ovary itself.
The embryo consists of the cotyledon(s) , epicotyl, and hypocotyl. The cotyledons resemble small leaves, and are usually the first photosynthetic organs of the plant. The portion of the embryo above the cotyledons is the epicotyl, and the portion below is the hypocotyl. The epicotyl is an apical meristem that produces the shoot of the growing plant and the first true leaves after germination. The hypocotyl develops into the root. Often the tip of the hypocotyl, the radicle, is the first indication of germination as it breaks out of the seed. Flowering plants are classified as monocotyledons or dicotyledons (most are now called eudicots ) based on the number of cotyledons produced in the embryo. Common monocotyledons include grasses, sedges, lilies, irises, and orchids; common dicotyledons include sunflowers, roses, legumes, snapdragons, and all nonconiferous trees.
The endosperm may be consumed by the embryo, as in many legumes, which use the cotyledons as a food source during germination. In other species the endosperm persists until germination, when it is used as a food
The Fruit
The fruit of a flowering plant is the mature ovary. As seeds mature, the surrounding ovary wall forms a protective structure that may aid in dispersal. The surrounding ovary tissue is called the pericarp and consists of three layers. From the outside to inside, these layers are the exocarp, mesocarp, and endocarp. The exocarp is usually tough and skinlike. The mesocarp is often thick, succulent, and sweet. The endocarp, which surrounds the seeds, may be hard and stony, as in most species with fleshy fruit, such as apricots.
A fruit is termed simple if it is produced by a single ripened ovary in a single flower (apples, oranges, apricots). An aggregated fruit is a cluster of mature ovaries produced by a single flower (blackberries, raspberries, strawberries). A multiple fruit is a cluster of many ripened ovaries on separate flowers growing together in the same inflorescence (pineapple, mulberry, fig). A simple fruit may be fleshy or dry. A fleshy simple fruit is classified as a berry (grape, tomato, papaya), pepo (cucumber, watermelon, pumpkin), hesperidium (orange), drupe (apricot), or pome (apple).
Dry simple fruits have a dry pericarp at maturity. They may or may not dehisce, or split, along a seam to release the seeds. A dehiscent dry fruit is classified as legume or pod (pea, bean), silique or silicle (mustard), capsule (poppy, lily), or follicle (milkweed, larkspur, columbine). An indehiscent dry fruit that does not split to release seeds is classified as an achene (sunflower, buttercup, sycamore), grain or caryopsis (grasses such as corn, wheat, rice, barley), schizocarp (carrot, celery, fennel), winged samara (maple, ash, elm), nut (acorn, chestnut, hazelnut), or utricle (duckweed family). Some fruiting bodies contain non-ovary tissue and are sometimes called pseudocarps. The sweet flesh of apples and pears, for example, is composed not of the pericarp but the receptacle, or upper portion, of the flowering shoot to which petals and other floral organs are attached.
Fruiting bodies of all kinds function to protect and disperse the seeds they contain. Protection can be physical (hard coverings) or chemical (repellents of seed predators). Sweet, fleshy fruits are attractive food for birds and mammals that consume seeds along with the fruit and pass the seeds intact in their fecal matter, which can act as a fertilizer. Dry fruits are usually adapted for wind dispersal of seeds, as for example with the assistance of winglike structures or a fluffy pappus that provides buoyancy. The diversity of fruiting bodies reflects in part the diversity of dispersal agents in the environment, which select for different fruit size, shape, and chemistry.
Pollination and Pollinators
Pollination is the movement of pollen from the stamens to the stigma, where germination and growth of the pollen tube occur. Most (approximately 96 percent) of all flowering plant species are hermaphroditic (possess both sexual functions within a plant, usually within every flower), and thus an individual can be pollinated by its own pollen or by pollen from another individual. Seed produced through self-pollination ("selfed" seed) is often inferior in growth, survival, and fecundity to seed produced through outcross pollination ("outcrossed" seed). As a result, in most species there is strong natural selection to maximize the proportion of outcrossed seed (the "outcrossing rate").
Flowering plants are unusual among seed plants in their superlative exploitation of animals (primarily insects) as agents of outcross pollination. The outcross pollination efficiency of insects, birds, and mammals (primarily bats) may have contributed to both the abundance and diversity of flowering plants. Abundance may have increased because of less wastage of energy and resources on unsuccessful pollen and ovules. Diversity may have increased for two reasons. First, insects undoubtedly have selected for a wide variety of floral forms that provide different rewards (pollen and nectar) and are attractive in appearance (color juxtaposition, size, shape) and scent (sweet, skunky) in different ways to different pollinators. Second, faithfulness of pollinators to particular familiar flowers may have reduced hybridization and speeded evolutionary divergence and the production of new species.
Although flowering plants first appeared after most of the major groups of insects had already evolved, flowering plants probably caused the evolution of many new species within these groups. Some new insect groups, such as bees and butterflies, originated after flowering plants, their members developing mouthpart structures and behavior specialized for pollination. In extreme cases, a plant is completely dependent on one insect species for pollination, and the insect is completely dependent on one plant species for food. Such tight interdependency occurs rarely but is well documented in yuccas/yucca moths, senita cacti/senita moths, and fig trees/some fig wasps. In all three insects, females lay eggs in the flowers, and their young hatch later to feed on the mature fruit and its contents. Females ensure that the fruit develops by gathering pollen from another plant and transporting it to the stigma of the flower holding their eggs. Plants benefit greatly in outcrossed seed produced, at the small cost of some consumed fruit and seeds, and the insects benefit greatly from the food supply for developing larvae at the small cost of transporting pollen the short distances between plants.
Pollinating agents, whether biotic or abiotic , have exerted strong selection on all aspects of the flower, resulting in the evolution of tremendous floral diversity. This diversity has been distilled into a small number of characteristic pollination syndromes.
Pollination by beetles selects usually for white color, a strong fruity scent, and a shallow, bowl-shaped flower. Bees select for yellow or blue/purple colorings, a landing platform with color patterns that guide the bee to nectar (often reflecting in the ultraviolet range of the spectrum), bilateral symmetry, and a sweet scent. Butterflies select for many colors other than yellow, a corolla (petal) tube with nectar at the base, and the absence of any scent. Moths in contrast select for nocturnally opening flowers with a strong scent and drab or white color, and also a tube with nectar at the base. Bats select also for nocturnally opening flowers, but with a strong musky scent and copious nectar, positioned well outside the foliage for easy access, and drab or white color. Hummingbirds select for red or orange flowers with no scent, copious nectar production, and a corolla tube with nectar at the base. Other pollinating birds that do not hover while feeding select for strong perches and flowers capable of containing copious nectar (tubes, funnels, cup shapes).

Wind as a pollinating agent selects for lack of color, scent, and nectar; small corolla; a large stigmatic surface area (usually feathery); abundantly produced, buoyant pollen; and usually erect styles and limp, hanging stamens. In addition there is great floral diversity within any of these syndromes, arising from the diverse evolutionary histories of the member plant species.
Selfing and Outcrossing
Most flowering plant species reproduce primarily by outcrossing, including the great majority of trees, shrubs, and perennial herbs. Adaptations that prevent self-fertilization include self-incompatibility (genetic recognition and blocking of self-pollen) and dioecy (separate male and female individuals). Adaptations that reduce the chances of self-pollination in hermaphrodites include separation of the anthers and stigma in space (herkogamy) or time (dichogamy). In many species, both self-incompatibility and spatiotemporal separation of the sex organs occur.
The ability to produce seeds by selfing, however, is advantageous in situations where outcrossing pollination is difficult or impossible. These include harsh environments where pollinators are rare or unpredictable, and regularly disturbed ground where survivors often end up isolated from each other. Selfing is also cheaper than outcrossing, because selfers can become pollinated without assistance from animals and therefore need not produce large, attractive flowers with abundant nectar and pollen rewards.
Most primarily selfing species are small annuals in variable or disturbed habitats, with small, drab flowers. Most desert annuals and roadside weeds, for example, are selfers. The evolutionary transition from outcrossing to near-complete selfing has occurred many times in flowering plants.
Outcrossing and selfing species differ in their evolutionary potential. Outcrossers are generally more genetically diverse and produce lineages that persist over long periods of evolutionary time, during which many new species are formed. Selfers, however, are less genetically diverse and tend to accumulate harmful mutations. They typically go extinct before they have an opportunity to evolve new species.
Asexual Reproduction
The ability to produce new individuals asexually is common in plants. Under appropriate experimental conditions, nearly every cell of a flowering plant is capable of regenerating the entire plant. In nature, new plants may be regenerated from leaves, stems, or roots that receive an appropriate stimulus and become separated from the parent plant. In most cases, these new plants arise from undifferentiated parenchyma cells, which develop into buds that produce roots and shoots before or after separating from the parent.
New plants can be produced from aboveground or belowground horizontal runners (stolons of strawberries, rhizomes of many grasses), tubers (potato, Jerusalem artichoke, dahlia), bulbs (onion, garlic), corms (crocus, gladiola), bulbils on the shoot (lily, many grasses), parenchyma cells in the leaves (Kalanchoe, African violet, jade plant) and inflorescence (arrowhead). Vegetative propagation is an economically important means of replicating valuable agricultural plants, through cuttings, layering, and grafting . Vegetative reproduction is especially common in aquatic vascular plants (for example, surfgrass and eelgrass), from which fragments can break off, disperse in the current, and develop into new whole plants.
A minority of flowering plants can produce seeds without the fusion of egg and sperm (known as parthenocarpy or agamospermy). This occurs when meiosis in the ovule is interrupted, and a diploid egg cell is produced, which functions as a zygote without fertilization. Familiar examples include citrus, dandelion, hawkweed, buttercup, blackberry/raspberry, and sorbus. Agamospermous species are more common at high elevations and at high latitudes, and nearly all have experienced a doubling of their chromosome number (tetraploidy) in their recent evolutionary history. These species experience evolutionary advantages and disadvantages similar to those of selfers.
Evolutionary Significance of Plant Reproduction Strategies
The attractive, colorful, and unique features of the most abundant and diverse group of land plants—the flowering plants—are believed to have evolved primarily to maximize the efficiency and speed of outcross reproduction. Each major burst of angiosperm evolution was a coevolutionary episode with associated animals, primarily insects, which were exploited to disperse pollen and seeds in ever more efficient and diverse ways.
The first major burst of flowering plant evolution was the appearance of the closed carpel together with showy flowers that were radially symmetrical . The closed carpel prevented self-fertilization through recognition and blocking of self pollen within the specialized conducting tissue of the style. Insects attracted to the showy flowers carried pollen between plants less wastefully than wind, and the radial symmetry accommodated insects of many sizes and shapes.
The second major burst was the appearance of bilaterally symmetrical flowers, which happened independently in many groups of plants at the same time that bees evolved. Bilateral symmetry forced bees to enter and exit flowers more precisely, promoting even more efficient outcross pollen transfer.
The third major burst of flowering plant evolution was the appearance of nutritious, fleshy fruits and seeds, coincident with a diversification of birds and rodents. The exploitation of vertebrates for fruit and seed dispersal resulted in less haphazard transport of offspring to neighboring populations of the same species (also visited as a food source), thereby reducing the chances that progeny inbreed with their siblings and parents and providing more assurance than wind currents that they find good habitat and unrelated mating partners of the same species.
SEE ALSO Alternation OF Generations ; Flowers ; Fruits ; Plant Nutrition ; Seeds
Stewart T. Schultz
Bibliography
Ayala, F. J., W. M. Fitch, and M. T. Clegg, eds. Variation and Evolution in Plants and Microorganisms. Washington, DC: National Academy Press, 2000.
Barth, F. G. Insects and Flowers. Princeton, NJ: Princeton University Press, 1991.
Baskin, C. C., and J. M. Baskin. Seeds: Ecology, Biogeography, and Evolution of Dormancy and Germination. San Diego, CA: Academic Press, 1998.
Briggs, D., and S. M. Walters. Plant Variation and Evolution, 3rd ed. New York: Cambridge University Press, 1997.
Chittka, L., and J. D. Thomson, eds. Cognitive Ecology of Pollination: Animal Behavior and Floral Evolution. New York: Cambridge University Press, 2001.
Dickison, W. C. Integrative Plant Anatomy. San Diego, CA: Harcourt/Academic Press, 2000.
Heywood, V. H., ed. Flowering Plants of the World. Oxford: Oxford University Press, 1993.
Marshall, C., and J. Grace. Fruit and Seed Production: Aspects of Development, Environmental Physiology and Ecology (Society for Experimental Biology Seminar, No. 47). New York: Cambridge University Press, 1992.
Proctor, M., P. Yeo, and A. Lack. The Natural History of Pollination. Portland, OR: Timber Press, 1996.
Richards, A. J. Plant Breeding Systems, 2nd ed. Cheltenham, England: Stanley Thornes Publishers Ltd., 1997.
Simpson, B. B., and M. Conner-Orgorzaly. Economic Botany: Plants in Our World, 2nd ed. New York: McGraw-Hill, 1995.